 Nitrogen is a chemical element with atomic number 7 which means there are 7 protons in its nucleus. 
Understanding these values is crucial for understanding the properties and behavior of atoms and the elements they make up.Atomic Number – Protons, Electrons and Neutrons in Nitrogen The mass number gives us information about the atom’s mass and stability, while the atomic number gives us information about its identity and electron configuration. In conclusion, an atom with a mass number of 13 and an atomic number of 6 has 7 neutrons in its nucleus. It is a synthetic element and has a very short half-life. Q: What is the heaviest element on the periodic table?Ī: The heaviest element on the periodic table is currently Oganesson, which has an atomic number of 118. 
This gives us the number of neutrons in the atom’s nucleus. Q: How do scientists determine the number of neutrons in an atom’s nucleus?Ī: Scientists determine the number of neutrons in an atom’s nucleus by subtracting the atomic number from the mass number. Isotopes are atoms of the same element with different numbers of neutrons in their nucleus. However, isotopes of the same element can have different mass numbers. Q: Can an atom have the same mass number but a different atomic number?Ī: No, each element has a unique atomic number, which defines its identity. This can result in the emission of particles or radiation, which can be harmful to living organisms. Atoms with too many or too few neutrons are unstable and tend to undergo radioactive decay. Q: What is the significance of the number of neutrons in an atom’s nucleus?Ī: The number of neutrons in an atom’s nucleus affects its stability and reactivity. Therefore, the atom has 7 neutrons in its nucleus. So, the number of neutrons in its nucleus is: In our example, the atom has a mass number of 13 and an atomic number of 6. Number of neutrons = Mass number – Atomic number To calculate the number of neutrons in an atom’s nucleus, we need to use the formula: How to Calculate the Number of Neutrons in an Atom’s Nucleus? Elements are defined by their atomic number, and each element has a unique atomic number. The atomic number gives us information about the atom’s identity and the number of electrons it has in its outer shell. It is denoted by the letter Z and is expressed as a whole number. The atomic number of an atom is the number of protons in its nucleus. This is because the nucleus becomes more crowded with protons and neutrons, increasing the likelihood of nuclear decay. The higher the mass number, the heavier the atom and the more unstable it is. The mass number gives us information about the atom’s mass and its stability. 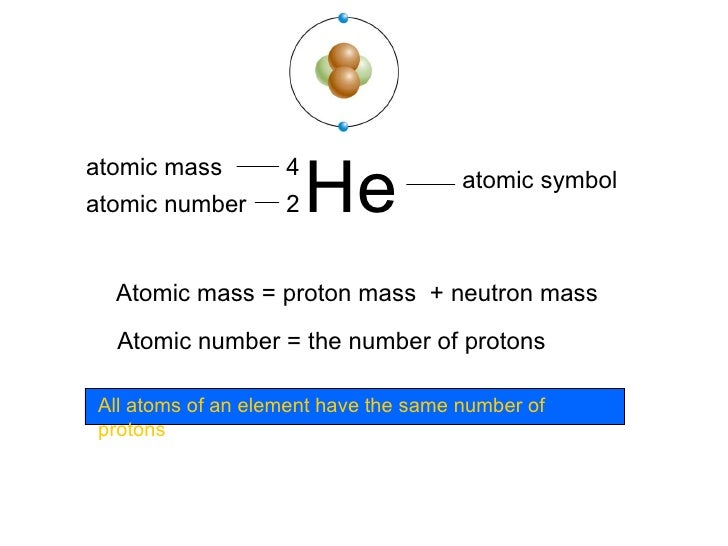
It is denoted by the letter A and is expressed in atomic mass units (amu). The mass number of an atom is the total number of protons and neutrons in its nucleus. We will learn how to calculate the number of neutrons in its nucleus and what this means for the atom’s properties. In this article, we will explore an atom with a mass number of 13 and an atomic number of 6. These two values give us important information about the atom’s properties, such as its identity and stability. When talking about atoms, we often hear about their mass number and atomic number. Understanding the Correlation Between Mass Number and Atomic Number in Neutron Calculation 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
